Prescription Required

Authentic Product

India's Leading Generic Pharmacy

Secure Payment
By NOVO NORDISK INDIA PVT LTD
MRP
₹
424.59
₹360.9
15 % OFF
Check Delivery
--
Content Reviewed By:
Dr. Sanjay Mehta
, (MBBS)
Written By:
Ms. Kavita Desai
, (B.Pharm)

Pregnancy
Generally safe under medical supervisionHuman insulin preparations such as Mixtard 30/70 are commonly used during pregnancy when insulin therapy is indicated, as maintaining tight glycaemic control is important for maternal and fetal outcomes.

BreastFeeding
Generally safeHuman insulin, including Mixtard 30/70, is considered compatible with breastfeeding, as it is a peptide that is broken down in the infant’s gastrointestinal tract and is not expected to cause harm at therapeutic maternal doses.

Alcohol
Avoid or limitAlcohol can unpredictably affect blood glucose levels and may increase the risk of delayed or prolonged hypoglycaemia in patients using Mixtard 30/70.

General Safety and Monitoring
Requires regular follow-upMixtard 30/70 therapy requires regular self-monitoring of blood glucose and periodic clinical review to assess glycaemic control, adjust doses, and screen for diabetes complications.
No, it isn't protected to drink liquor while utilizing Mixtard 30 HM 100IU/ml Penfill. Drinking liquor while utilizing this medication might bring down your glucose levels and cause startling hypoglycemia.
Keep this prescription in the compartment or the pack it came in, immovably shut. Store it as demonstrated by the rules referred to on the pack or name. Discard the unused medication. Ensure it isn't consumed by kids and others.
Mixtard 30 HM 100IU/ml Penfill is to be infused under the skin (subcutaneously). The best regions to give Mixtard 30 HM 100IU/ml Penfill are your stomach, upper thigh, upper arm or bottom.
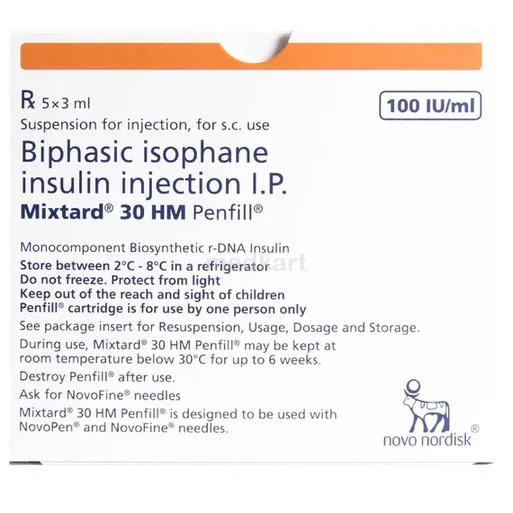
Mixtard 30/70 is a biphasic human insulin preparation used to control blood glucose in people with diabetes mellitus. It contains 30% soluble (regular) human insulin and 70% isophane (NPH) human insulin in a suspension. The soluble component provides relatively rapid onset of action to cover meal-related glucose rises, while the isophane component provides an intermediate, longer-lasting effect to cover basal insulin needs.
Mixtard 30/70 is prescribed for the treatment of diabetes mellitus, including type 1 diabetes and type 2 diabetes when oral antidiabetic medicines alone are insufficient or not appropriate. It is used to improve glycaemic control and reduce the risk of diabetes-related complications as part of a comprehensive management plan that includes diet, physical activity, and regular blood glucose monitoring.
Mixtard 30/70 works by replacing or supplementing the insulin that the body either does not produce at all or does not use effectively. The 30% soluble insulin component starts to act relatively soon after injection and helps control the rise in blood glucose that occurs after meals. The 70% isophane insulin component is absorbed more slowly and provides a longer duration of action, helping to maintain baseline (fasting and between-meal) blood glucose levels. Together, they provide both prandial and basal insulin coverage.
Mixtard 30/70 is administered by subcutaneous injection only, typically into the fatty tissue of the abdomen, thigh, upper arm, or buttock. Injection sites should be rotated within the same region to reduce the risk of lipodystrophy or skin thickening. The suspension should be gently resuspended as per the product instructions before injection. It must not be injected into a vein and should only be used with appropriate insulin delivery devices as instructed by a healthcare professional.
The dose and timing of Mixtard 30/70 must be individualized and determined by a healthcare professional based on blood glucose measurements, diet, physical activity, and other medications. Patients should not make major dose changes or alter the injection schedule on their own. Minor adjustments may sometimes be made according to a plan previously agreed with the treating clinician, but any persistent changes in blood glucose control, illness, or lifestyle should prompt medical review before altering the regimen.
The most common side effect of Mixtard 30/70 is hypoglycaemia (low blood glucose), which can present with symptoms such as sweating, shakiness, palpitations, hunger, headache, irritability, confusion, or visual disturbances. Local injection-site reactions such as redness, swelling, itching, or mild pain can occur, usually transiently. Long-term use at the same injection site may cause lipodystrophy or skin thickening. Allergic reactions are less common but can range from local reactions to, rarely, generalized hypersensitivity.
If you miss a scheduled dose of Mixtard 30/70, you should follow the advice provided by your healthcare professional or diabetes care plan. In general, you should not take a double dose to make up for a missed injection, as this increases the risk of hypoglycaemia. Check your blood glucose, consider when you last ate, and contact your healthcare provider or diabetes nurse for individualized guidance, especially if you are unsure how to proceed or if your blood glucose is significantly elevated.
Mixtard 30/70 can be used alone or in combination with certain oral antidiabetic medicines, such as metformin or other agents, depending on the individual treatment plan. The decision to combine insulin with oral medicines is made by the treating clinician based on the type of diabetes, degree of glycaemic control, comorbidities, and risk of hypoglycaemia. Any changes in concomitant therapy require close monitoring of blood glucose and possible adjustment of insulin doses by a healthcare professional.
Unopened Mixtard 30/70 should generally be stored in a refrigerator at the temperature range specified in the product information, protected from freezing and direct heat. Once in use, the penfill or cartridge is usually kept at room temperature within the recommended range and used within the period stated in the package leaflet. The product should not be frozen or exposed to excessive heat or direct sunlight. Always check the appearance of the suspension and do not use it if there are clumps, discoloration, or visible particles that do not disperse after gentle mixing.
Mixtard 30/70 should not be used by individuals with known hypersensitivity to human insulin or any of the excipients in the formulation. It must not be used in episodes of hypoglycaemia. Caution and specialist supervision are required in people with conditions that significantly alter insulin requirements, such as severe liver or kidney impairment, endocrine disorders, or acute illness. It is also not appropriate for people who are unable to recognize or respond to hypoglycaemia without adequate support.
Switching from another insulin preparation to Mixtard 30/70 should only be done under medical supervision. Differences in insulin type, concentration, timing of action, and individual sensitivity mean that close monitoring of blood glucose is required during and after the switch. The healthcare professional will determine the appropriate conversion approach and may adjust the total daily insulin requirement, injection timing, and monitoring frequency to maintain safe and effective glycaemic control.
Good service and affordable price I think best in medical
Pradeep Singh Rathore
•
Reviewed on 05-11-2022
(5/5)
Very responsive staff.All drugs available at store
Ronak Makwana
•
Reviewed on 16-01-2024
(5/5)
The customer care was ans the response to customer was fabulo
sagar sonagra
•
Reviewed on 17-01-2024
(5/5)
Interactive and knowledgeable
Naval Kava
•
Reviewed on 01-04-2024
(5/5)
Nice service All required drugs are available 😊
Meet Dobariya
•
Reviewed on 13-01-2024
(5/5)
NOVO NORDISK INDIA PVT LTD
Country of Origin -
India

MRP
₹
424.59
₹360.9
15 % OFF
Quick Links
Medkart's sole intention is to ensure that its consumers get information that is expert-reviewed, accurate and trustworthy. However, the information contained herein should NOT be used as a substitute for the advice of a qualified physician. The information provided here is for informational purposes only. This may not cover everything about particular health conditions, medicines, generic alternatives, all possible side effects, drug interactions, warnings, alerts, lab tests, etc. Please consult your doctor and discuss all your queries related to any disease or medicine. We intend to support, not replace, the doctor-patient relationship.
10 Lakh+
Happy customers
35000+
Pin-codes Covered
75 Lakh+
Orders Delivered

Authentic Products
All WHO-GMP Certified Medicines
©2025 Medkart Pharmacy. All Rights Reserved